CHEMISTRY 112 / U15
Section 201
Prof. Andrew Pounds, Ph.D.
Office: Rm. 105 Willett Science Center, (478) 301-5627
e-mail: pounds_aj@mercer.edu
Alt. Phone (SMS) : (478) 227-3444
Office Hours: M 1:00 - 3:00 p.m., R 1:00 - 2:00 p.m. (or by appointment)
CHM 112 is the second course in a two-part sequence to introduce
students to the foundational principles of Chemistry. Students
in CHM 112 will be exposed to the fundamental laws of mass and
energy conservation and their application to chemical systems and
reactions. Students will also study the role of energy and entropy
in chemical
systems and how they manifest themselves in chemical equilibrium.
The approach to equilibrium will also be investigated through
the study of chemical kinetics.
Students in CHM 112 are expected to
read at the college level and also set up and solve algebraic and
trigonometric equations. As such students must have satisfied the
math prerequisites for CHM 111 and made a grade of C or better
in CHM 111.
Upon completion of this course,
a student will demonstrate competence in each of the following areas:
- solving chemical problems,
- understanding chemical concepts from the lecture by
successfully applying these concepts on homework and tests,
- making accurate physical and chemical measurements in the
laboratory, and
- writing clear and concise laboratory reports.
Class Meeting Times and Locations
Lecture: MTWR 10:15 a.m. - 12:15 p.m., WSC LEC
Lab: TW 1:00 - 4:00 p.m., WSC 302
Course Materials
Chemistry, Julia Burdge, 3 ed.,
ed.,
Sapling Key for Online Homework
Laboratory Notebook (numbered, carbonless pages, available at bookstore)
Scientific Calculator (TI or HP with Numerical Equation Solver highly recommended)
Laboratory Instructions (From WWW)
Approved Laboratory Safety Glasses/Goggles and Clothing
Lock (for lab drawer)
Course Structure
Nine chapters of the text will be covered during the semester in
the order listed on the class schedule. The lecture time will be
used to expound on and augment the text and also discuss problem
solving strategies. Students are responsible for all material
covered in class as well as the material from the textual sections
listed in the class schedule. While homework will not be collected,
students are encouraged to complete as many problems as possible to
gain competency with the material. Several unannounced in-class
quizzes will be given during the semester. The best eight grades
from the quizzes will count toward the final grade.
Four 50 minute exams will be given per the schedule and the best
three exam grades will count toward the final grade. A three hour
final exam will be administered at the end of the term.
Grading
| Tests ( best 3 @ 100 pts) |
300 pts |
| Quizzes (best 6 @ 25 pts) |
150 pts |
| Online Sapling Homework |
50 pts |
| Laboratory (8 @ 30 pts) |
240 pts |
| Final Exam |
260 pts |
| |
|
| Total Possible |
1000 pts |
The following grading scale is assured but may be slightly lowered based on test results.
A 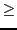 900 pts
900 pts
B 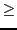 800 pts
800 pts
C 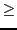 700 pts
700 pts
D 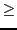 600 pts
600 pts
F  600 pts
600 pts
General Information
Honor Code: All students in CHM 112 are expected to adhere
to the Mercer University Honor Code. Any suspected violations will
be reported to the Honor Council for further investigation.
Attendance: Except for the first day of the semester,
attendance will not be taken. However, students are still
accountable for all material covered in class as well as any
announcements made during the lecture period. Attendance is
required in lab. See policies in the lab section.
Missed Quizzes: No makeup quizzes will be given.
Missed Exams: Anyone missing an exam for any reason
(personal illness, death in the immediate family, or other emergency)
must notify Dr. Pounds in advance. The absence will be considered
unexcused otherwise. Make-up exams will be individually scheduled.
Partial Credit: Partial credit will not be awarded on any
quiz, exam, prelab, or lab report unless individuals show their
work and clearly delineate how they arrived at their answers.
Re-grading Policy: If a student suspects that an error was
made in the grading of a submitted work, they may return the paper
for re-grading with the understanding that the entire work will be
re-graded and not only the portion in question.
Posting of Grades: Grades will not be posted
throughout the semester.
If you have questions,
see Dr. Pounds.
E-mail Listserve: I maintain an e-mail listserve
which I use use copiously to send information to the class and which you
can use to communicate
with each other. To sign up for the listserve and to learn how to
send information to it, please go to:
http://theochem.mercer.edu/mailman/listinfo/chm112.
American Disability Act:
``Students with a documented disability must inform the instructor at the
close of the first class meeting. The instructor will refer you to the
Student Support Services office for consultation regarding evaluation,
documentation of your disability, and a recommendation as to the
accommodation, if any, to be provided. Students must provide instructors
with an accommodation form from Student Support Services listing reasonable
accommodation to sign and return to Student Support Services. The Student
Support Services office is located on the 3rd floor of the Conned Student
Center.
If you do NOT consult with the instructor and follow up at the Student
Support Services office during the first two weeks of classes, as provided
above, you will thereby waive any claim to a disability and the right to
any accommodation pertaining thereto.''
Electronic Submission of Materials: ``Students bear sole
responsibility for ensuring that papers or assignments submitted
electronically to a professor are received in a timely manner and
in the electronic format(s) specified by the professor. Students
are therefore obliged to have their e-mail client issue a receipt
verifying that the document has been received. Students are also
strongly advised to retain a copy of the dated submission on a
separate disk. Faculty members are encouraged, but not required,
to acknowledge receipt of the assignment.''
Sapling Learning Online Homework:
Online homework assignments will be posted at saplinglearning.com, which you must purchase. Begin the assignments early so that you have an opportunity to address any technology issues. Late work will not be accepted except in extreme circumstances. Homework is an individual assignment; it is an honor code violation to work in groups or ask others how to do problems. As a group, you can study problems from the text on the same topics; however, when you are actually doing the online homework, it should reflect your individual understanding.
Directions for Students to Access Sapling:
- Go to Sapling Learning and click on "US Higher Ed" at the top right.
If you already have an account skip to step 3 below.
- If you do not have an account
- If you have a Facebook account, you can use it to quickly create a Sapling Learning account.
Click ``Create an Account'', then ``Create my account through Facebook''. You will be prompted
to log into Facebook if you aren't already. Choose a username and password, then click
``Link Account''. You can then skip to step 3.
- Otherwise, you can click ``Create an Account''. Supply the requested information
and click ``Create My Account''. Check your email (and spam filter) for a message
from Sapling Learning and click on the link provided in that email.
- Find your course in the list (you may need to expand the subject and term categories)
and click the link (Mercer University - CHM 112 - Summer15 - POUNDS). (Of course,
this will change based on what your course is called.)
- Select a payment option and following the remaining instructions.
Once you have registered and enrolled, you can log in at any time
to complete or review your homework assignments. During sign up or
throughout the term, if you have any technical problems or grading
issues, send an email to support@saplinglearning.com explaining the
issue. The Sapling Learning support team is almost always faster
and better able to resolve issues than your instructor.
The bookstore also has ISBN numbers which will allow those on
scholarship to have their sapling paid by their scholarship.
They should have these numbers from my summer course, but if not,
the Tech TA (or Devin Lindsey - manager) can supply you with those
ISBN numbers.
Tips for Succeeding in Chemistry 112
There is unquestionably a lot of material to be covered in Chemistry
112. It is also more mathematically rigorous than CHM 111.
For that reason it is imperative to keep up with the class.
The last thing you want to worry about is covering two chapters of
new material the night before the test. You are expected to keep
up with the class reading and problems. The unannounced quizzes
are an added incentive for you to do this.
The book contains numerous problems which you should attemp. In
addition I will give you a selection of problems you should attempt.
The book problems and the additional problems
are representative of some of the problems you will see on the
exams. To become proficient you will need practice. In short,
work as many problems as you can before the test. The student
solutions manual provides solutions to the odd numbered problems.
In addition, solutions to other problems
described in class will sometimes be posted on the
WWW (http://theochem.mercer.edu/chm112), or worked in class.
Students who need solutions to additional even-numbered problems
should contact Dr. Pounds. While the aforementioned problems
are not required, you are strongly encouraged to complete them.
You are required to complete the online homework as your performance
on these problems will be a part of the overall grade for the course.
For more help in the course you should utilize the SI sessions
for your class as well as the University's free tutoring service.
Both of these resources are here for you to use and, although
they can not guarantee a higher grade in the course, they will
most likely increase your understanding of chemistry and thereby
positively affect your performance in the class.
Tentative Class Schedule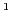
| Week Starting |
Chapter Sections |
Lecture and Problem Solving Topics |
June 28 |
18.1 - 18.5 |
Energy and Entropy (Review) |
| |
10.1 - 10.7 |
Properties of Gases |
| |
|
Derivation of the Ideal Gas Law |
| |
|
Gas Mixtures and Reactions of Gases |
| |
|
Kinetic Theory of Gases |
| |
|
Real Gases and Intermolecular Forces |
| |
11.1 - 11.7 |
Properties of Liquids |
| |
|
Vapor Pressure |
| |
|
Crystal Structure and Lattices |
| |
|
Properties of Solids |
| |
|
Phase Changes, Phase Diagrams |
| |
13.1 - 13.7 |
Physical Properties of Solutions |
| |
|
Concentration and Solubility |
| |
|
Colligative Properties |
| |
|
Colloids |
July 5 |
|
EXAM #1, 7/6/15 |
| |
14.1 - 14.6 |
Reaction Rates, Rate Laws and Reaction Orders |
| |
|
Integrated Rate Laws |
| |
|
Temperature Dependence |
| |
|
Reaction Mechanisms, Catalysis |
July 12 |
|
EXAM #2, 7/13/15 |
| |
15.1 - 15.5 |
Principles of Equilibria |
| |
|
Equilibrium Constants |
| |
18.6 |
Themodynamics and Equilibrium |
| |
|
Equilibrium Expression and The Law of Mass Action |
| |
|
Solving Difficult Equilibrium Problems |
| |
|
Le Châtelier's Principle |
July 19 |
|
EXAM #3, 7/20/15 |
| |
|
LAST DAY FOR COURSE WITHDRAWAL 7/20/15 |
| |
16.1 - 16.12 |
Acid/Base Theories |
| |
|
Self-Ionization of Water and pH |
| |
|
Weak Acids, Ionization Constants |
| |
|
Acid-Base Conjugate Pairs |
| |
|
Polyprotic Acids |
| |
|
Molecular Structure and Acid Strength |
| |
|
Hydrolysis |
| |
17.1 - 17.6 |
The Common Ion Effect |
| |
|
Acid/Base Equilibrium |
| |
|
Buffers and Titrations |
| |
|
Solubility Equilibria |
| |
|
Common Ions and Solubility |
| |
|
Complex Ions |
| |
|
Precipitation Reactions |
July 26 |
|
EXAM #4, 7/27/15 |
| |
19.1 - 19.6 |
Galvanic Cells |
| |
|
Cell Potentials |
| |
|
Free Energy, Equilibrium, and Cell Potential |
| |
|
The Nernst Equation |
| |
|
Batteries and Corrosion |
| |
|
FINAL EXAM, 7/31/15, 1 p.m. |
| |
|
(Friday) |
 I reserve the right
to modify this schedule as situations warrant.
I reserve the right
to modify this schedule as situations warrant.
The Laboratory
Safety always comes first in lab. Developing good lab safety
habits is important, even if the day's lab activities are not
particularly dangerous. You will not be allowed in lab if
you are not prepared. That means being appropriately dressed,
having your safety glasses and knowing what you are supposed
to do during the lab. The lab schedule and instructions are available at:
http://chemistry.mercer.edu/genchem/chm112.htm
The lab manual pdfs SHOULD NOT be printed and brought to lab.
No points will be awarded
for printed laboratory procedures. Instead, read the manual and think
about what you are going to do and why. Write down the procedure and
any questions you have in your lab notebook before coming to lab.
If
you do not have your notebook with the hand written procedure in
it, you won't be allowed in lab. The lab report forms available
from the same web site should be printed and turned in along with
the yellow copies from your notebook. Data and observations MUST
to be written in your notebook, not on the lab report form.
No credit is available for
the lab report if you miss lab for any unexcused reason, including
showing up unprepared, or if you are more than 10 minutes late. It is
important to show up on time, since we will go over safety notes in
the first few minutes. You will lose 1 pt for each safety violation
in a lab period (ie. removal of safety glasses for any reason in
the laboratory). Lab reports are due before, NOT during the next
laboratory session.
It is imperative that you attend lab. During the summer session
it is impossible to make up a lab.
If you must miss a laboratory meeting for a Mercer University event,
you need to see Dr. Pounds at least one week in advance for remediation
purposes.
Students will not be
penalized for laboratories missed due to excused absence (as defined
below) and a complete
laboratory report will still be required on the announced due date.
A passing grade for CHM 112 will NOT be available to any
student who misses three or more laboratory meetings (excused
or otherwise).

Laboratory Grading:
There will be eight pre-lab exercises (5 pts each) and a eight lab reports
(25 pts each) over the course of the semester
for a total of 240 pts. The laboratory component
is thus worth 24% of your final grade for
CHM 112. Labs are due at the beginning of class on
Tuesday of each week.
Late labs will
lose 10 points/day, with a grade of zero resulting if the write-up
is more than 2 days late.
Prelabs:
Pre-lab exercises will be administered via BlackBoard.
More information related to these exercises will be provided prior to the first
lab via e-mail.
Laboratory Policies:
Cell Phone: Out of courtesy for all those participating in the learning experience, all cell phones and pagers must be turned off before entering any classroom, lab, or formal academic or performance event.
Chemical Sensitivity Statement: This course includes the
handling of chemicals, and the reasonable accommodation policy also
applies to any chemical sensitivity, allergy, or other physical
or medical condition that might limit a student's ability to
participate in the required course activities. In these cases,
the instructor may require a physician's documentation of the
student's condition before arranging accommodation. If the
instructor determines that the student's condition cannot be
reasonably accommodated, then the student will be asked to select
an alternative course.
Laboratory Schedule
| Lab Day |
Experiment |
June 30 |
Check-In |
| |
Molar Mass of a Volatile Liquid |
July 1 |
Freezing Point Depression |
July 7 |
Concentration Kinetics |
July 8 |
Temperature Kinetics |
July 14 |
Gas Equilibrium |
July 15 |
Equilibrium in Aqueous Solution |
July 21 |
Buffers |
July 22 |
Thermodynamics of Borax Solubility |
| |
Check-Out |
This document was generated using the
LaTeX2HTML translator Version 2008 (1.71)
Copyright © 1993, 1994, 1995, 1996,
Nikos Drakos,
Computer Based Learning Unit, University of Leeds.
Copyright © 1997, 1998, 1999,
Ross Moore,
Mathematics Department, Macquarie University, Sydney.
The command line arguments were:
latex2html -split 0 -local_icons -no_navigation syl_u15.tex
The translation was initiated by Andrew J. Pounds on 2015-06-20
Andrew J. Pounds
2015-06-20
